
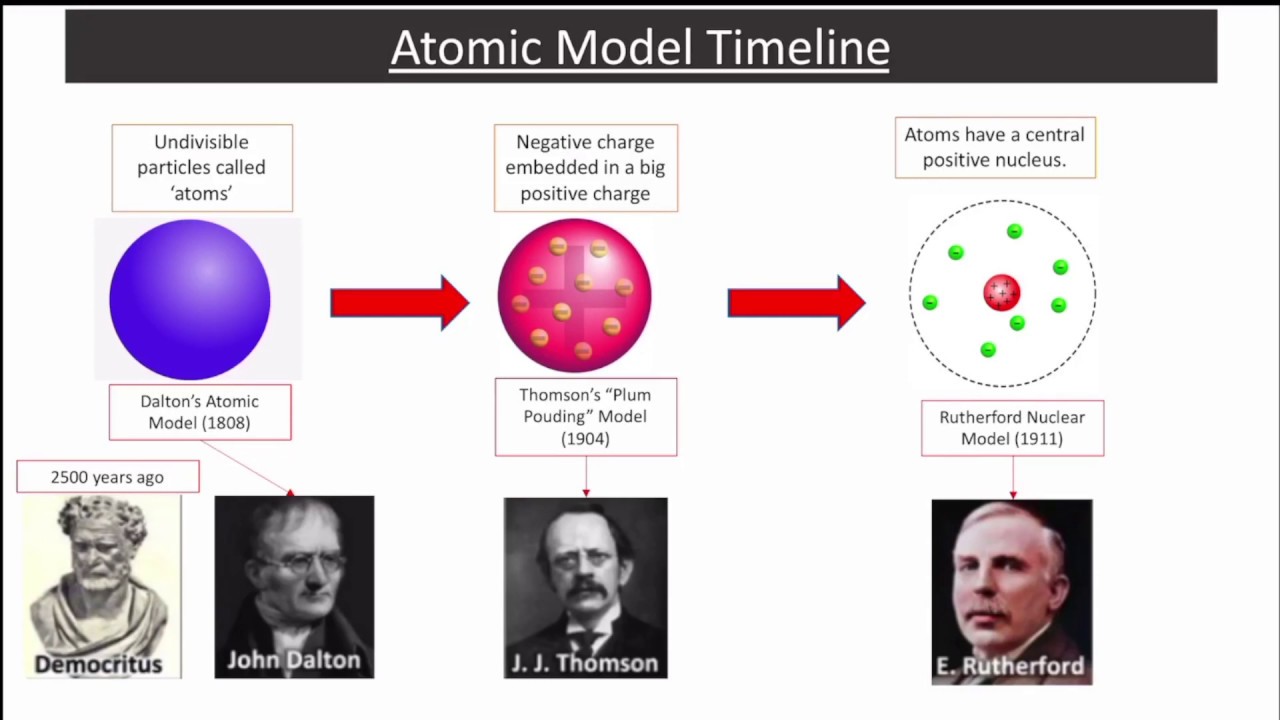
Another scenario allowed the electrons to orbit at right-angles to the plane of the rings, resulting in a series of concentric shells of electrons. For example, one description has the electrons uniformly spaced and travelling “at high speed” in a series of concentric “rings” (i.e., orbits), with progressively more electrons in each ring as distance from the atomic centre increases. In fact, Thomson’s 1904 description of the model4 contains concepts that foreshadow both the Bohr-Rutherford model and Gilbert Lewis’ electron-dot symbolism. The “plum pudding” analogy also fails to adequately represent Thomson’s model.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
